Artificial Intelligence (AI) is rapidly transforming many sectors of healthcare, and one of the most significant areas of impact is pharmacovigilance. Pharmacovigilance refers to the science and activities related to detecting, assessing, understanding, and preventing adverse effects or other drug-related problems. Traditionally, pharmacovigilance relied heavily on manual processes, data review by safety professionals, and retrospective analysis. However, with the explosion of healthcare data, traditional systems often struggle to keep pace.
This is where AI in pharmacovigilance plays a critical role. AI technologies such as machine learning, natural language processing (NLP), and advanced analytics can process massive datasets quickly and identify potential drug safety signals faster than traditional approaches.
At the same time, the use of AI in drug safety systems introduces regulatory challenges, workforce changes, and global policy considerations. Regulatory authorities are working to develop frameworks that ensure innovation does not compromise patient safety.
In this article, we explore how artificial intelligence is shaping pharmacovigilance through regulatory guidance, global adoption trends, and workforce transformation.
AI in Pharmacovigilance

The integration of AI into pharmacovigilance is transforming how pharmaceutical companies monitor drug safety. Modern pharmacovigilance systems are expected to handle large volumes of adverse event reports from multiple sources, including clinical trials, healthcare professionals, patient reports, social media, and electronic health records.
Traditional manual systems often struggle with this scale of information. AI technologies help automate repetitive tasks, detect patterns in safety data, and improve decision-making in drug safety monitoring.
The growing adoption of AI in pharmacovigilance allows pharmaceutical companies and healthcare regulators to detect safety risks earlier, reduce processing time, and improve patient safety outcomes.
Navigating the FDA’s AI Framework
Regulatory compliance is one of the most important considerations when implementing AI technologies in healthcare. Regulatory agencies such as the U.S. Food and Drug Administration (FDA) have started developing frameworks that guide the safe use of AI in healthcare and drug safety monitoring.
These frameworks aim to ensure that AI-based systems are reliable, transparent, and safe for public health.
What are Regulatory Expectations

Regulators emphasize several key principles when evaluating AI systems in pharmacovigilance:
- Transparency in algorithms
- Data quality and reliability
- Traceability of decision-making processes
- Continuous monitoring of AI performance
Pharmaceutical companies must demonstrate that AI systems used for safety monitoring operate within strict quality standards.
Challenges in Regulatory Compliance
Implementing AI in pharmacovigilance is not straightforward. Regulatory authorities expect companies to validate their AI models thoroughly. Companies must also demonstrate that automated systems can be audited and reviewed.
This creates a challenge because many AI models, especially deep learning systems, function as “black boxes,” making it difficult to explain how certain decisions are made.
Balancing Innovation and Safety
Regulatory frameworks aim to balance innovation with patient protection. While AI has the potential to revolutionize drug safety monitoring, regulators want to ensure that technology does not introduce new risks.
Pharmaceutical companies must therefore work closely with regulatory authorities to develop AI systems that are both innovative and compliant.
The Promise of AI in Pharmacovigilance
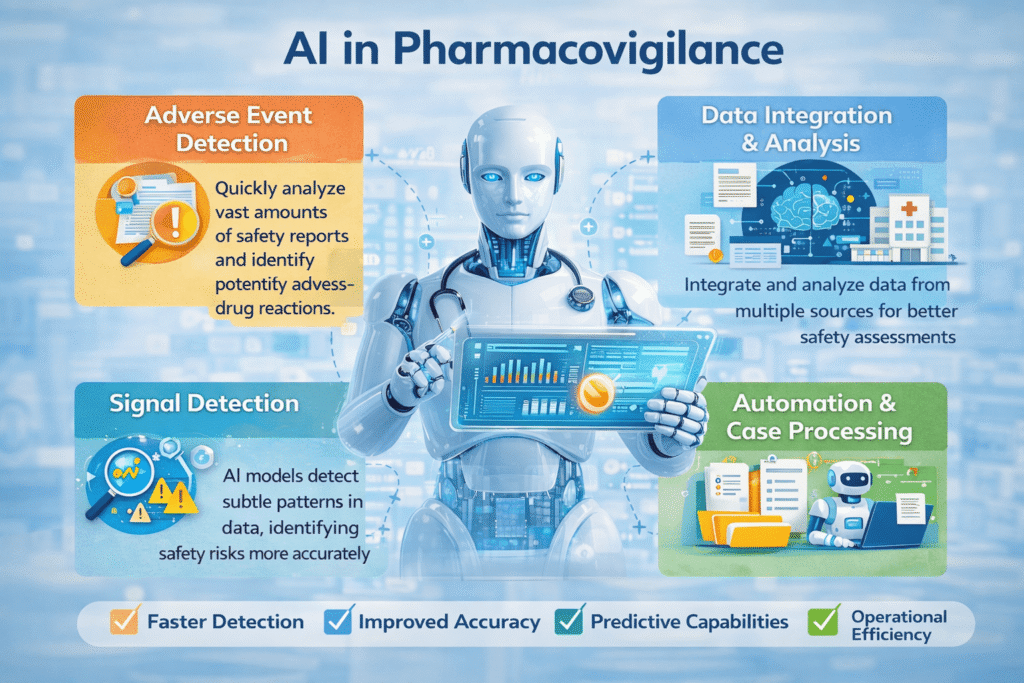
The adoption of AI in pharmacovigilance offers several transformative benefits for the healthcare industry. These benefits go beyond simple automation and include improvements in accuracy, speed, and predictive capabilities.
Faster Detection of Adverse Events
AI can analyze millions of safety reports in a fraction of the time required by manual systems. Natural language processing tools can scan medical literature, social media posts, and patient reports to identify potential adverse drug reactions.
This allows companies to detect safety signals earlier and respond more quickly.
Improved Data Processing
Pharmacovigilance systems receive information from multiple sources, including hospitals, healthcare providers, regulatory submissions, and patient feedback. AI algorithms can integrate and analyze these diverse datasets efficiently.
This improves the quality of safety assessments and helps organizations identify trends that might otherwise remain hidden.
Enhanced Signal Detection
Signal detection refers to identifying patterns that suggest a possible relationship between a drug and an adverse event. AI models can detect subtle patterns in large datasets that human analysts might miss.
Machine learning algorithms continuously improve over time as they process more data, increasing their effectiveness in identifying safety risks.
Automation of Routine Tasks
Pharmacovigilance teams often spend significant time performing repetitive tasks such as data entry, case processing, and report classification. AI-powered automation can handle many of these activities, allowing safety professionals to focus on more complex analytical tasks.
The Need for Specificity in Guidance
While AI offers many benefits, regulatory guidance around its implementation in pharmacovigilance remains evolving. One of the key challenges faced by pharmaceutical companies is the lack of highly specific guidelines for AI-based safety monitoring systems.
Ambiguity in Current Regulations
Existing regulatory frameworks often provide high-level principles rather than detailed instructions. While this flexibility allows innovation, it also creates uncertainty for organizations implementing AI technologies.
Companies may struggle to determine:
- How AI models should be validated
- What level of transparency is required
- How AI decision-making processes should be documented
Importance of Clear Standards
More specific guidance would help companies adopt AI technologies with greater confidence. Clear regulatory standards could define best practices for:
- AI model validation
- Data governance
- Risk management
- Performance monitoring
This would reduce compliance risks while encouraging innovation.
Ensuring Responsible AI Adoption
Specific regulatory guidance also ensures that AI systems are implemented responsibly. Clear rules can help prevent issues such as algorithm bias, data privacy violations, and inaccurate safety assessments.
The Evolving Role of the Emerging Drug Safety Technology Program
Technology-driven drug safety programs are becoming increasingly important as pharmacovigilance evolves. These initiatives aim to encourage innovation while maintaining strong safety standards.
Supporting Innovation in Drug Safety
Drug safety technology programs provide a structured environment where pharmaceutical companies can test and evaluate new technologies.
These programs help regulators understand how emerging tools such as AI, automation, and advanced analytics can be used in pharmacovigilance systems.
Collaboration Between Industry and Regulators
One of the most important aspects of these programs is collaboration. Pharmaceutical companies, regulatory authorities, and technology developers work together to explore new approaches to drug safety monitoring.
This collaboration helps regulators develop policies that reflect real-world technological capabilities.
Accelerating Technological Adoption
By supporting research and pilot projects, drug safety technology programs help accelerate the adoption of AI-driven pharmacovigilance systems.
These initiatives ensure that technological innovation continues to advance without compromising patient safety.
Global Perspectives on AI in Safety Monitoring
The adoption of AI in pharmacovigilance is not limited to a single region. Countries around the world are exploring how artificial intelligence can improve drug safety monitoring.
However, regulatory approaches and adoption levels vary significantly between regions.
Adoption Across Different Healthcare Systems
Many developed healthcare systems are investing heavily in AI-driven pharmacovigilance technologies. Governments and healthcare organizations recognize the potential of AI to improve safety monitoring and reduce operational costs.
In contrast, developing countries often face challenges related to infrastructure, data availability, and technical expertise.
Differences in Regulatory Approaches
Different countries have different regulatory frameworks governing AI in healthcare. Some regulators are more proactive in developing AI guidelines, while others are still evaluating potential policies.
This creates challenges for multinational pharmaceutical companies that must comply with multiple regulatory systems.
Need for Global Collaboration
Global collaboration is essential to harmonize pharmacovigilance practices. International organizations and regulatory bodies are working to establish shared standards for AI-based safety monitoring.
Harmonized regulations could simplify compliance and improve global patient safety.
Expanding Access in Underserved Regions
One of the most promising aspects of AI in pharmacovigilance is its potential to expand drug safety monitoring in underserved regions.
Addressing Infrastructure Challenges
Many developing countries lack robust pharmacovigilance infrastructure. AI-powered systems can help overcome these limitations by automating safety monitoring processes.
For example, AI tools can analyze patient data from mobile health platforms and identify potential safety issues.
Improving Drug Safety Monitoring
In regions with limited healthcare resources, AI technologies can help identify adverse drug reactions more efficiently. Automated systems reduce the need for large teams of manual reviewers.
This makes pharmacovigilance more scalable and accessible.
Enhancing Global Patient Safety
Expanding pharmacovigilance capabilities in underserved regions improves global patient safety. Better safety monitoring ensures that medications remain safe and effective for diverse populations worldwide.
Reimagining the Pharmacovigilance Workforce
The rise of AI is also transforming the workforce responsible for drug safety monitoring.
Changing Skill Requirements
Traditional pharmacovigilance roles focused primarily on manual data review and case processing. However, the integration of AI technologies requires new skill sets.
Professionals now need expertise in:
- Data analytics
- Machine learning concepts
- Healthcare informatics
- Digital health technologies
Training and Reskilling Professionals
Organizations must invest in training programs that help pharmacovigilance professionals adapt to new technologies. Continuous learning will be essential for staying relevant in an AI-driven environment.
Human-AI Collaboration
AI is not replacing pharmacovigilance professionals. Instead, it is augmenting their capabilities.
AI systems handle repetitive tasks and data processing, while human experts provide clinical judgment, regulatory oversight, and strategic decision-making.
This collaboration between humans and AI creates a more efficient and effective drug safety ecosystem.
Conclusion
Artificial intelligence is rapidly transforming the landscape of pharmacovigilance. The integration of AI in pharmacovigilance enables faster detection of safety signals, improved data processing, and more efficient monitoring of drug safety.
However, the adoption of AI also introduces challenges related to regulatory compliance, workforce transformation, and global policy alignment.
As regulatory frameworks continue to evolve, pharmaceutical companies must work closely with authorities to ensure responsible AI implementation. At the same time, investments in workforce development and technological infrastructure will be essential for maximizing the benefits of AI-driven safety monitoring.
Ultimately, the combination of advanced technology, regulatory oversight, and human expertise will shape the future of pharmacovigilance and ensure safer medicines for patients worldwide.
FAQs
What is AI in pharmacovigilance?
AI in pharmacovigilance refers to the use of artificial intelligence technologies such as machine learning and natural language processing to monitor drug safety, detect adverse events, and improve pharmacovigilance processes.
Why is AI important for drug safety monitoring?
AI enables faster processing of large datasets, improves signal detection accuracy, and automates routine pharmacovigilance tasks. This helps pharmaceutical companies identify potential safety risks more efficiently.
How do regulators evaluate AI systems in pharmacovigilance?
Regulatory authorities evaluate AI systems based on factors such as data quality, transparency, reliability, validation processes, and compliance with safety guidelines.
Can AI replace pharmacovigilance professionals?
No. AI supports pharmacovigilance professionals by automating repetitive tasks and analyzing large datasets. Human experts remain essential for clinical interpretation, regulatory decisions, and safety oversight.
What is the future of AI in pharmacovigilance?
The future of AI in pharmacovigilance includes greater use of predictive analytics, real-time safety monitoring, global regulatory harmonization, and improved collaboration between human experts and intelligent systems.
Debabrata Behera is a passionate blogger who writes about digital trends, personal growth, and practical insights, helping readers stay informed, inspired, and ready to achieve success in life.